 Indian medical students might be flocking to Chinese universities to study medicine due to lack of opportunities back home, but Indian small and medium scale bulk drug manufacturers are finding quality to be a major set back in procurements from China.
Indian medical students might be flocking to Chinese universities to study medicine due to lack of opportunities back home, but Indian small and medium scale bulk drug manufacturers are finding quality to be a major set back in procurements from China.
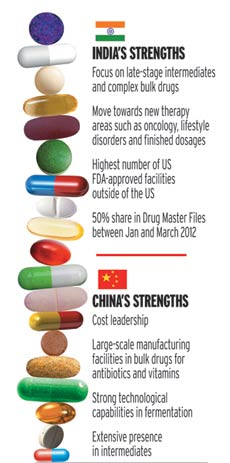
China and India are the leading players in bulk drugs or active pharmaceutical ingredients (APIs) which are used to make medicines. Both countries account for more than 40 percent of global bulk drug production. China is the largest bulk drug supplier, and India is second. China is a key supplier of bulk drugs to India – exporting 60 percent of India’s US$7 billion bulk drug imports, however with few quality controls, sans inspections and dismal regulations, China’s low costing bulk drugs are no longer incentive enough for Indian pharmaceutical manufacturers. Further, the Indian bulk drug market is valued at US$13.5 billion, and its Chinese counterpart at US$30 billion.
India has already dealt with the problem of fake ‘Made in India‘ drugs being sold in Africa, now poor quality API are causing an increasing number of Indian phrama companies to manufacture their own bulk drugs in India.
Shasun Chemicals and Drugs, a Chennai-based Rs 1,000-crore company, has decided to manufacture some intermediates at its Andhra Pradesh plant, rather than importing them from China. Managing Director S. Abhaya Kumar says: “We have earmarked Rs 70 crore to move production of some of our key intermediates to India. The shift should be complete by December”, he told Business Today. Shasun, the largest producer of ibuprofen in the world adds that besides quality, intermediate prices in China have also increased by 10 percent in the last three months, due to rising petroleum prices and pressure on China to adhere to environmental norms. Shasun is developing 13 APIs for a few billion-dollar drugs whose patents expire in the next five years.
Overhauling their phrama sectors has become crucial to both China and India, and it is here that India has an advantage. While India’s price points are a lot higher, international quality norms, and inspections are adhered to. Both countries have spent billions of dollars in research and development, manufacturing bulk drugs to replace the those whose patents expire in the next five years. While health concerns reign in both China and India, it is the government which needs to step in and crank in controls across the board. In India, New Delhi needs to put better processes in place, so that cheaper, bad quality Chinese drugs don’t flood Indian markets, and aren’t used to manufacture drugs, they also need to create strong financials so they can invest in better infrastructure and move up the value chain. In China, Beijing needs to adhere to international quality standards, put processes in places for regular inspections and make sure quality rather than low cost drugs are exported.
